Interferon gamma Antibody - #DF6045
| Product: | Interferon gamma Antibody |
| Catalog: | DF6045 |
| Description: | Rabbit polyclonal antibody to Interferon gamma |
| Application: | WB IHC |
| Cited expt.: | WB, IHC |
| Reactivity: | Human, Mouse, Rat |
| Mol.Wt.: | 19-25kDa; 19kD(Calculated). |
| Uniprot: | P01579 |
| RRID: | AB_2838015 |
Product Info
*The optimal dilutions should be determined by the end user. For optimal experimental results, antibody reuse is not recommended.
*Tips:
WB: For western blot detection of denatured protein samples. IHC: For immunohistochemical detection of paraffin sections (IHC-p) or frozen sections (IHC-f) of tissue samples. IF/ICC: For immunofluorescence detection of cell samples. ELISA(peptide): For ELISA detection of antigenic peptide.
Cite Format: Affinity Biosciences Cat# DF6045, RRID:AB_2838015.
Fold/Unfold
IFG; IFI; IFN gamma; IFN, immune; IFN-gamma; IFNG; IFNG_HUMAN; Immune interferon; Interferon gamma;
Immunogens
A synthesized peptide derived from human Interferon gamma, corresponding to a region within the internal amino acids.
- P01579 IFNG_HUMAN:
- Protein BLAST With
- NCBI/
- ExPASy/
- Uniprot
MKYTSYILAFQLCIVLGSLGCYCQDPYVKEAENLKKYFNAGHSDVADNGTLFLGILKNWKEESDRKIMQSQIVSFYFKLFKNFKDDQSIQKSVETIKEDMNVKFFNSNKKKRDDFEKLTNYSVTDLNVQRKAIHELIQVMAELSPAAKTGKRKRSQMLFRGRRASQ
Research Backgrounds
Produced by lymphocytes activated by specific antigens or mitogens. IFN-gamma, in addition to having antiviral activity, has important immunoregulatory functions. It is a potent activator of macrophages, it has antiproliferative effects on transformed cells and it can potentiate the antiviral and antitumor effects of the type I interferons.
Proteolytic processing produces C-terminal heterogeneity, with proteins ending alternatively at Gly-150, Met-157 or Gly-161.
Secreted.
Released primarily from activated T lymphocytes.
Belongs to the type II (or gamma) interferon family.
Research Fields
· Cellular Processes > Cell growth and death > Necroptosis. (View pathway)
· Environmental Information Processing > Signaling molecules and interaction > Cytokine-cytokine receptor interaction. (View pathway)
· Environmental Information Processing > Signal transduction > HIF-1 signaling pathway. (View pathway)
· Environmental Information Processing > Signal transduction > TGF-beta signaling pathway. (View pathway)
· Environmental Information Processing > Signal transduction > Jak-STAT signaling pathway. (View pathway)
· Genetic Information Processing > Folding, sorting and degradation > Proteasome.
· Human Diseases > Endocrine and metabolic diseases > Type I diabetes mellitus.
· Human Diseases > Infectious diseases: Bacterial > Salmonella infection.
· Human Diseases > Infectious diseases: Parasitic > Leishmaniasis.
· Human Diseases > Infectious diseases: Parasitic > Chagas disease (American trypanosomiasis).
· Human Diseases > Infectious diseases: Parasitic > African trypanosomiasis.
· Human Diseases > Infectious diseases: Parasitic > Malaria.
· Human Diseases > Infectious diseases: Parasitic > Toxoplasmosis.
· Human Diseases > Infectious diseases: Parasitic > Amoebiasis.
· Human Diseases > Infectious diseases: Bacterial > Tuberculosis.
· Human Diseases > Infectious diseases: Viral > Measles.
· Human Diseases > Infectious diseases: Viral > Influenza A.
· Human Diseases > Infectious diseases: Viral > Herpes simplex infection.
· Human Diseases > Infectious diseases: Viral > Epstein-Barr virus infection.
· Human Diseases > Cancers: Overview > Pathways in cancer. (View pathway)
· Human Diseases > Immune diseases > Inflammatory bowel disease (IBD).
· Human Diseases > Immune diseases > Systemic lupus erythematosus.
· Human Diseases > Immune diseases > Rheumatoid arthritis.
· Human Diseases > Immune diseases > Allograft rejection.
· Human Diseases > Immune diseases > Graft-versus-host disease.
· Organismal Systems > Development > Osteoclast differentiation. (View pathway)
· Organismal Systems > Immune system > Antigen processing and presentation. (View pathway)
· Organismal Systems > Immune system > Natural killer cell mediated cytotoxicity. (View pathway)
· Organismal Systems > Immune system > IL-17 signaling pathway. (View pathway)
· Organismal Systems > Immune system > Th1 and Th2 cell differentiation. (View pathway)
· Organismal Systems > Immune system > Th17 cell differentiation. (View pathway)
· Organismal Systems > Immune system > T cell receptor signaling pathway. (View pathway)
References
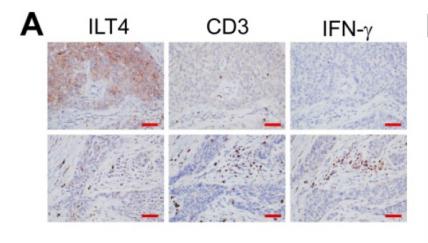
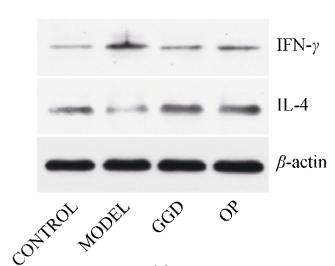
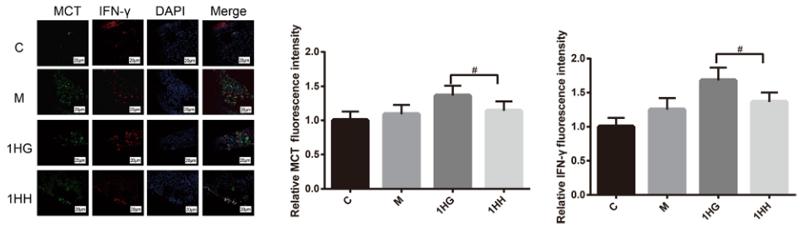
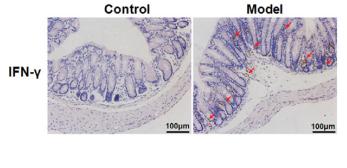
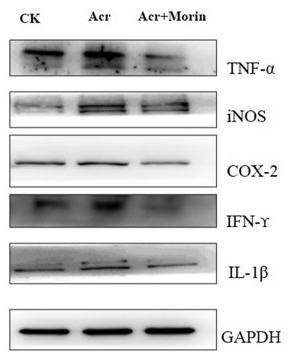
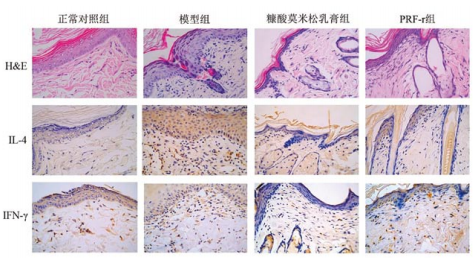
Application: IHC Species: Mouse Sample: colonic tissues
Application: IHC Species: human Sample: tumor cells
Restrictive clause
Affinity Biosciences tests all products strictly. Citations are provided as a resource for additional applications that have not been validated by Affinity Biosciences. Please choose the appropriate format for each application and consult Materials and Methods sections for additional details about the use of any product in these publications.
For Research Use Only.
Not for use in diagnostic or therapeutic procedures. Not for resale. Not for distribution without written consent. Affinity Biosciences will not be held responsible for patent infringement or other violations that may occur with the use of our products. Affinity Biosciences, Affinity Biosciences Logo and all other trademarks are the property of Affinity Biosciences LTD.