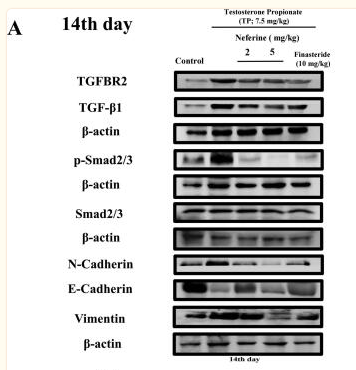
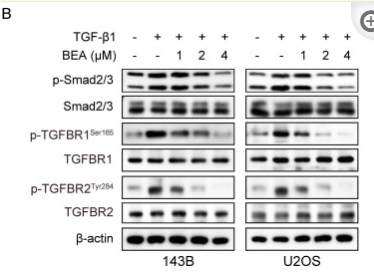
TGFBR2 Antibody - #AF5449
| Product: | TGFBR2 Antibody |
| Catalog: | AF5449 |
| Description: | Rabbit polyclonal antibody to TGFBR2 |
| Application: | WB IHC |
| Cited expt.: | WB, IHC |
| Reactivity: | Human, Mouse, Rat |
| Prediction: | Pig, Bovine, Horse, Rabbit, Dog, Chicken, Xenopus |
| Mol.Wt.: | 63 kDa; 65kD(Calculated). |
| Uniprot: | P37173 |
| RRID: | AB_2837933 |
Product Info
*The optimal dilutions should be determined by the end user. For optimal experimental results, antibody reuse is not recommended.
*Tips:
WB: For western blot detection of denatured protein samples. IHC: For immunohistochemical detection of paraffin sections (IHC-p) or frozen sections (IHC-f) of tissue samples. IF/ICC: For immunofluorescence detection of cell samples. ELISA(peptide): For ELISA detection of antigenic peptide.
Cite Format: Affinity Biosciences Cat# AF5449, RRID:AB_2837933.
Fold/Unfold
AAT3; FAA3; LDS1B; LDS2; LDS2B; MFS2; RIIC; TAAD2; TbetaR II; TbetaR-II; TGF beta receptor type 2; TGF beta receptor type II; TGF beta receptor type IIB; TGF beta type II receptor; TGF-beta receptor type II; TGF-beta receptor type-2; TGF-beta type II receptor; TGF-beta-R2; TGFB R2; TGFbeta - RII; TGFbeta RII; Tgfbr2; TGFR-2; TGFR2_HUMAN; Transforming growth factor beta receptor II; Transforming growth factor beta receptor type II; Transforming growth factor beta receptor type IIC; Transforming growth factor, beta receptor II (70/80kDa); transforming growth factor, beta receptor II alpha; transforming growth factor, beta receptor II beta; transforming growth factor, beta receptor II delta; transforming growth factor, beta receptor II epsilon; transforming growth factor, beta receptor II gamma; Transforming growth factor-beta receptor type II;
Immunogens
A synthesized peptide derived from human TGFBR2, corresponding to a region within the internal amino acids.
- P37173 TGFR2_HUMAN:
- Protein BLAST With
- NCBI/
- ExPASy/
- Uniprot
MGRGLLRGLWPLHIVLWTRIASTIPPHVQKSVNNDMIVTDNNGAVKFPQLCKFCDVRFSTCDNQKSCMSNCSITSICEKPQEVCVAVWRKNDENITLETVCHDPKLPYHDFILEDAASPKCIMKEKKKPGETFFMCSCSSDECNDNIIFSEEYNTSNPDLLLVIFQVTGISLLPPLGVAISVIIIFYCYRVNRQQKLSSTWETGKTRKLMEFSEHCAIILEDDRSDISSTCANNINHNTELLPIELDTLVGKGRFAEVYKAKLKQNTSEQFETVAVKIFPYEEYASWKTEKDIFSDINLKHENILQFLTAEERKTELGKQYWLITAFHAKGNLQEYLTRHVISWEDLRKLGSSLARGIAHLHSDHTPCGRPKMPIVHRDLKSSNILVKNDLTCCLCDFGLSLRLDPTLSVDDLANSGQVGTARYMAPEVLESRMNLENVESFKQTDVYSMALVLWEMTSRCNAVGEVKDYEPPFGSKVREHPCVESMKDNVLRDRGRPEIPSFWLNHQGIQMVCETLTECWDHDPEARLTAQCVAERFSELEHLDRLSGRSCSEEKIPEDGSLNTTK
Predictions
Score>80(red) has high confidence and is suggested to be used for WB detection. *The prediction model is mainly based on the alignment of immunogen sequences, the results are for reference only, not as the basis of quality assurance.
High(score>80) Medium(80>score>50) Low(score<50) No confidence
Research Backgrounds
Transmembrane serine/threonine kinase forming with the TGF-beta type I serine/threonine kinase receptor, TGFBR1, the non-promiscuous receptor for the TGF-beta cytokines TGFB1, TGFB2 and TGFB3. Transduces the TGFB1, TGFB2 and TGFB3 signal from the cell surface to the cytoplasm and is thus regulating a plethora of physiological and pathological processes including cell cycle arrest in epithelial and hematopoietic cells, control of mesenchymal cell proliferation and differentiation, wound healing, extracellular matrix production, immunosuppression and carcinogenesis. The formation of the receptor complex composed of 2 TGFBR1 and 2 TGFBR2 molecules symmetrically bound to the cytokine dimer results in the phosphorylation and the activation of TGFRB1 by the constitutively active TGFBR2. Activated TGFBR1 phosphorylates SMAD2 which dissociates from the receptor and interacts with SMAD4. The SMAD2-SMAD4 complex is subsequently translocated to the nucleus where it modulates the transcription of the TGF-beta-regulated genes. This constitutes the canonical SMAD-dependent TGF-beta signaling cascade. Also involved in non-canonical, SMAD-independent TGF-beta signaling pathways.
Phosphorylated on a Ser/Thr residue in the cytoplasmic domain.
Cell membrane>Single-pass type I membrane protein. Membrane raft.
Belongs to the protein kinase superfamily. TKL Ser/Thr protein kinase family. TGFB receptor subfamily.
Research Fields
· Cellular Processes > Transport and catabolism > Endocytosis. (View pathway)
· Cellular Processes > Cell growth and death > Cellular senescence. (View pathway)
· Cellular Processes > Cellular community - eukaryotes > Adherens junction. (View pathway)
· Environmental Information Processing > Signal transduction > MAPK signaling pathway. (View pathway)
· Environmental Information Processing > Signaling molecules and interaction > Cytokine-cytokine receptor interaction. (View pathway)
· Environmental Information Processing > Signal transduction > FoxO signaling pathway. (View pathway)
· Environmental Information Processing > Signal transduction > TGF-beta signaling pathway. (View pathway)
· Environmental Information Processing > Signal transduction > Hippo signaling pathway. (View pathway)
· Human Diseases > Infectious diseases: Parasitic > Chagas disease (American trypanosomiasis).
· Human Diseases > Infectious diseases: Viral > HTLV-I infection.
· Human Diseases > Cancers: Overview > Pathways in cancer. (View pathway)
· Human Diseases > Cancers: Overview > Transcriptional misregulation in cancer.
· Human Diseases > Cancers: Specific types > Colorectal cancer. (View pathway)
· Human Diseases > Cancers: Specific types > Pancreatic cancer. (View pathway)
· Human Diseases > Cancers: Specific types > Chronic myeloid leukemia. (View pathway)
· Human Diseases > Cancers: Specific types > Hepatocellular carcinoma. (View pathway)
· Human Diseases > Cancers: Specific types > Gastric cancer. (View pathway)
· Organismal Systems > Development > Osteoclast differentiation. (View pathway)
· Organismal Systems > Immune system > Th17 cell differentiation. (View pathway)
· Organismal Systems > Endocrine system > Relaxin signaling pathway.
References
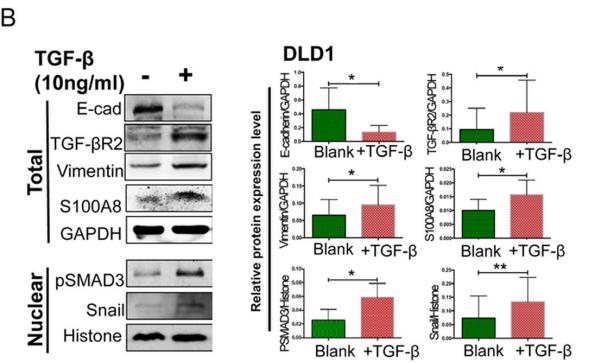
Application: WB Species: human Sample: DLD1 cells
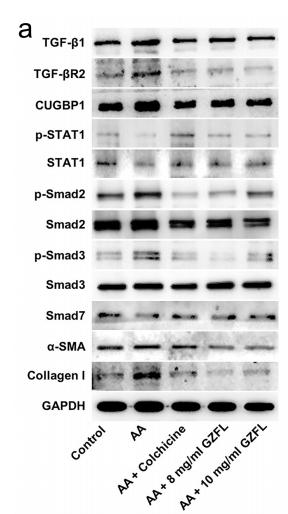
Application: WB Species: human Sample: NPC cells
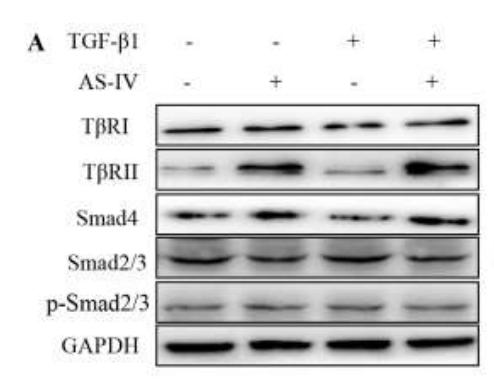
Application: WB Species: Mouse Sample: OS cells
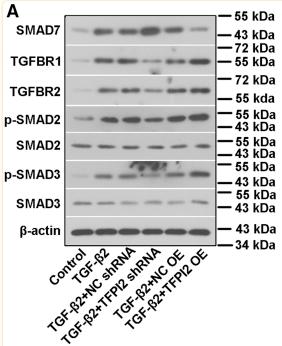
Application: WB Species: human Sample: hPDLSCs
Application: IHC Species: pig Sample:
Application: WB Species: human Sample: LX-2 cells
Application: WB Species: Human Sample: hRGECs
Application: WB Species: human Sample: SW962 cells
Restrictive clause
Affinity Biosciences tests all products strictly. Citations are provided as a resource for additional applications that have not been validated by Affinity Biosciences. Please choose the appropriate format for each application and consult Materials and Methods sections for additional details about the use of any product in these publications.
For Research Use Only.
Not for use in diagnostic or therapeutic procedures. Not for resale. Not for distribution without written consent. Affinity Biosciences will not be held responsible for patent infringement or other violations that may occur with the use of our products. Affinity Biosciences, Affinity Biosciences Logo and all other trademarks are the property of Affinity Biosciences LTD.