Calnexin Antibody - #AF5362
| Product: | Calnexin Antibody |
| Catalog: | AF5362 |
| Description: | Rabbit polyclonal antibody to Calnexin |
| Application: | WB IHC IF/ICC |
| Cited expt.: | WB |
| Reactivity: | Human, Mouse, Rat, Monkey |
| Prediction: | Pig, Zebrafish, Bovine, Horse, Sheep, Rabbit, Dog, Chicken, Xenopus |
| Mol.Wt.: | 90 kDa; 68kD(Calculated). |
| Uniprot: | P27824 |
| RRID: | AB_2837847 |
Related Downloads
Protocols
Product Info
*The optimal dilutions should be determined by the end user. For optimal experimental results, antibody reuse is not recommended.
*Tips:
WB: For western blot detection of denatured protein samples. IHC: For immunohistochemical detection of paraffin sections (IHC-p) or frozen sections (IHC-f) of tissue samples. IF/ICC: For immunofluorescence detection of cell samples. ELISA(peptide): For ELISA detection of antigenic peptide.
Cite Format: Affinity Biosciences Cat# AF5362, RRID:AB_2837847.
Fold/Unfold
Calnexin; CALX_HUMAN; CANX; CNX; FLJ26570; Histocompatibility complex class I antigen binding protein p88; IP90; Major histocompatibility complex class I antigen-binding protein p88; p90;
Immunogens
A synthesized peptide derived from human Calnexin, corresponding to a region within C-terminal amino acids.
- P27824 CALX_HUMAN:
- Protein BLAST With
- NCBI/
- ExPASy/
- Uniprot
MEGKWLLCMLLVLGTAIVEAHDGHDDDVIDIEDDLDDVIEEVEDSKPDTTAPPSSPKVTYKAPVPTGEVYFADSFDRGTLSGWILSKAKKDDTDDEIAKYDGKWEVEEMKESKLPGDKGLVLMSRAKHHAISAKLNKPFLFDTKPLIVQYEVNFQNGIECGGAYVKLLSKTPELNLDQFHDKTPYTIMFGPDKCGEDYKLHFIFRHKNPKTGIYEEKHAKRPDADLKTYFTDKKTHLYTLILNPDNSFEILVDQSVVNSGNLLNDMTPPVNPSREIEDPEDRKPEDWDERPKIPDPEAVKPDDWDEDAPAKIPDEEATKPEGWLDDEPEYVPDPDAEKPEDWDEDMDGEWEAPQIANPRCESAPGCGVWQRPVIDNPNYKGKWKPPMIDNPSYQGIWKPRKIPNPDFFEDLEPFRMTPFSAIGLELWSMTSDIFFDNFIICADRRIVDDWANDGWGLKKAADGAAEPGVVGQMIEAAEERPWLWVVYILTVALPVFLVILFCCSGKKQTSGMEYKKTDAPQPDVKEEEEEKEEEKDKGDEEEEGEEKLEEKQKSDAEEDGGTVSQEEEDRKPKAEEDEILNRSPRNRKPRRE
Predictions
Score>80(red) has high confidence and is suggested to be used for WB detection. *The prediction model is mainly based on the alignment of immunogen sequences, the results are for reference only, not as the basis of quality assurance.
High(score>80) Medium(80>score>50) Low(score<50) No confidence
Research Backgrounds
Calcium-binding protein that interacts with newly synthesized glycoproteins in the endoplasmic reticulum. It may act in assisting protein assembly and/or in the retention within the ER of unassembled protein subunits. It seems to play a major role in the quality control apparatus of the ER by the retention of incorrectly folded proteins. Associated with partial T-cell antigen receptor complexes that escape the ER of immature thymocytes, it may function as a signaling complex regulating thymocyte maturation. Additionally it may play a role in receptor-mediated endocytosis at the synapse.
Phosphorylated at Ser-564 by MAPK3/ERK1. phosphorylation by MAPK3/ERK1 increases its association with ribosomes (By similarity).
Palmitoylation by DHHC6 leads to the preferential localization to the perinuclear rough ER. It mediates the association of calnexin with the ribosome-translocon complex (RTC) which is required for efficient folding of glycosylated proteins.
Ubiquitinated, leading to proteasomal degradation. Probably ubiquitinated by ZNRF4.
Endoplasmic reticulum membrane>Single-pass type I membrane protein. Endoplasmic reticulum. Melanosome.
Note: Identified by mass spectrometry in melanosome fractions from stage I to stage IV (PubMed:12643545, PubMed:17081065). The palmitoylated form preferentially localizes to the perinuclear rough ER (PubMed:22314232).
Belongs to the calreticulin family.
Research Fields
· Cellular Processes > Transport and catabolism > Phagosome. (View pathway)
· Genetic Information Processing > Folding, sorting and degradation > Protein processing in endoplasmic reticulum. (View pathway)
· Human Diseases > Infectious diseases: Viral > HTLV-I infection.
· Organismal Systems > Immune system > Antigen processing and presentation. (View pathway)
· Organismal Systems > Endocrine system > Thyroid hormone synthesis.
References
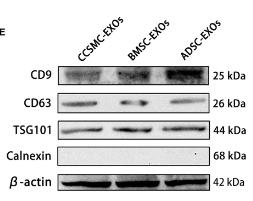
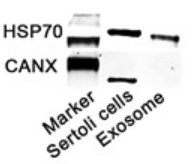
Application: WB Species: Human Sample:
Application: WB Species: Mice Sample: Tumour cells
Application: WB Species: Human Sample:
Application: WB Species: Mouse Sample:
Restrictive clause
Affinity Biosciences tests all products strictly. Citations are provided as a resource for additional applications that have not been validated by Affinity Biosciences. Please choose the appropriate format for each application and consult Materials and Methods sections for additional details about the use of any product in these publications.
For Research Use Only.
Not for use in diagnostic or therapeutic procedures. Not for resale. Not for distribution without written consent. Affinity Biosciences will not be held responsible for patent infringement or other violations that may occur with the use of our products. Affinity Biosciences, Affinity Biosciences Logo and all other trademarks are the property of Affinity Biosciences LTD.