GFP-tag Antibody - #T0005
Product Info
Source:
Mouse
Application:
WB 1:3000-1:10000, IHC 1:50-1:200, IF/ICC: 1:100-1:500, IP 1:100-1:500
*The optimal dilutions should be determined by the end user. For optimal experimental results, antibody reuse is not recommended.
*Tips:
*The optimal dilutions should be determined by the end user. For optimal experimental results, antibody reuse is not recommended.
*Tips:
WB: For western blot detection of denatured protein samples. IHC: For immunohistochemical detection of paraffin sections (IHC-p) or frozen sections (IHC-f) of tissue samples. IF/ICC: For immunofluorescence detection of cell samples. ELISA(peptide): For ELISA detection of antigenic peptide.
Reactivity:
All
Clonality:
Monoclonal [5F67]
Specificity:
GFP-Tag Mouse Monoclonal antibody detects endogenous levels of C-terminal, internal, and N-terminal GFP-tagged proteins.
RRID:
AB_2839413
Cite Format: Affinity Biosciences Cat# T0005, RRID:AB_2839413.
Cite Format: Affinity Biosciences Cat# T0005, RRID:AB_2839413.
Conjugate:
Unconjugated.
Purification:
Affinity-chromatography.
Storage:
Mouse IgG1 in phosphate buffered saline (without Mg2+ and Ca2+), pH 7.4, 150mM NaCl, 0.02% sodium azide and 50% glycerol. Store at -20 °C. Stable for 12 months from date of receipt.
Immunogens
Immunogen:
Full-length GFP protein.
Description:
N/A
References
1). Discovery of a potent allosteric activator of DGKQ that ameliorates obesity-induced insulin resistance via the sn-1,2-DAG-PKCε signaling axis. Cell metabolism, 2020
(PubMed: 36525963)
[IF=27.7]
2). AGGF1 therapy inhibits thoracic aortic aneurysms by enhancing integrin α7-mediated inhibition of TGF-β1 maturation and ERK1/2 signaling. Nature communications, 2023
(PubMed: 37081014)
[IF=16.6]
3). CPT1A induction following epigenetic perturbation promotes MAVS palmitoylation and activation to potentiate antitumor immunity. Molecular cell, 2023
(PubMed: 38016475)
[IF=14.5]
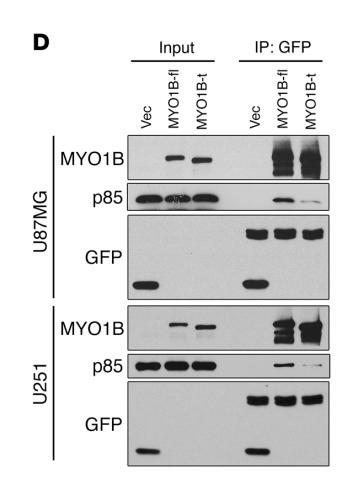
4). Splicing factor SRSF1 promotes gliomagenesis via oncogenic splice-switching of MYO1B. The Journal of Clinical Investigation, 2019
(PubMed: 30481162)
[IF=13.3]
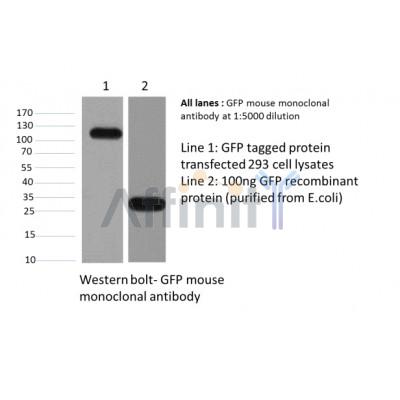
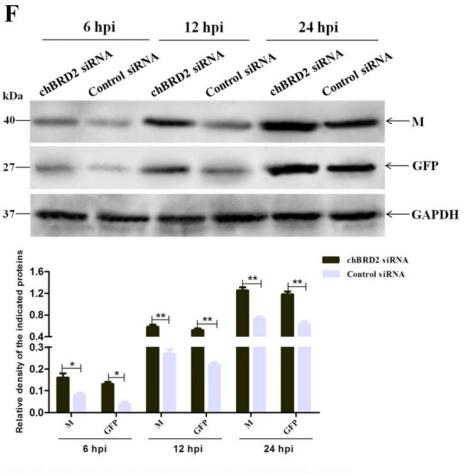
Application: WB Species: human Sample: U87MG and U251 cells
Figure 10.| SRSF1-guided MYO1B splicing determines cell fate through the PDK1/AKT and PAK/LIMK pathways. (D) Co-IP confirmation of the interaction between EGFP-fused MYO1B proteins (MYO1B-fl and MYO1B-t) and endogenous p85 PI3K in U87MG and U251 cells.
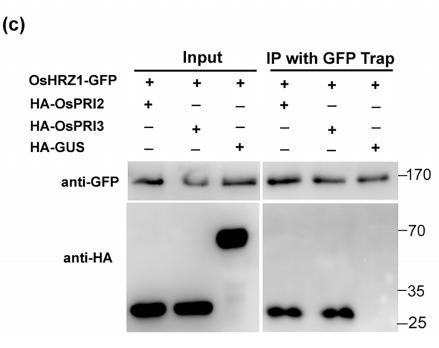
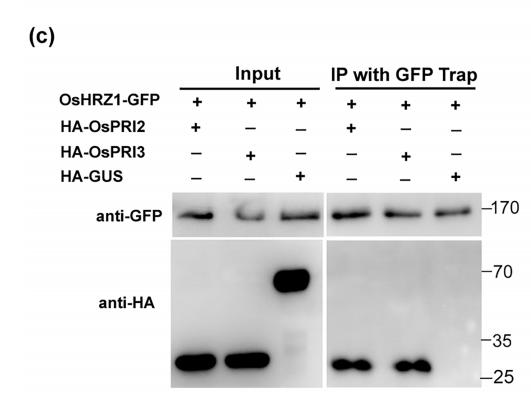
5). Oryza sativa POSITIVE REGULATOR OF IRON DEFICIENCY RESPONSE 2 (OsPRI2) and OsPRI3 are involved in the maintenance of Fe homeostasis. PLANT CELL AND ENVIRONMENT, 2020
(PubMed: 31674679)
[IF=6.0]
Application: WB Species: yeast Sample: yeast cells
FIGURE 1| Interaction of OsHRZ1 with OsPRI2 and OsPRI3.(c) CoIP assay. Total proteins from different combinations with OsHRZ1‐GFP and HA‐OsPRI2/HA‐OsPRI3/HA‐GUS were immunoprecipitated with GFP‐Trap followed by immunoblotting with the indicated antibodies. HRZ1‐GFP/HA‐GUS, negative control. Protein molecular weight (in kDa) is indicated.
Application: WB Species: Plant Sample: Root
FIGURE 1 Interaction of OsHRZ1 with OsPRI2 and OsPRI3. (a) Yeast two‐hybrid assays. Yeast co‐transformed with different BD and AD
plasmid combinations were spotted in parallel in 10‐fold dilution series on synthetic dropout medium lacking Leu/Trp/His/Ade. The C‐terminal
truncated OsHRZ1 and full‐length OsPRI2/3 were cloned into pGBKT7 and pGADT7, respectively. OsHRZ1‐C/OsPRI1, positive control. OsHRZ1‐
C/Empty, negative control. (b) Pull‐down assay. OsHRZ1 was fused with the GST tag and OsPRI2/3 were fused with the His tag. Recombinant
proteins were expressed in E. coli. Proteins were pulled down by glutathione Sepharose 4B and detected using the anti‐His antibody. Protein
molecular weight (in kDa) is indicated. (c) CoIP assay. Total proteins from different combinations with OsHRZ1‐GFP and HA‐OsPRI2/HA‐OsPRI3/
HA‐GUS were immunoprecipitated with GFP‐Trap followed by immunoblotting with the indicated antibodies. HRZ1‐GFP/HA‐GUS, negative
control. Protein molecular weight (in kDa) is indicated. (d) Degradation of OsPRI2 or OsPRI3 was carried out by detecting the OsPRI2/3‐GFP
protein level in co‐infiltration experiments with increasing amounts of OsHRZ1‐GFP. GFP proteins were used as an internal control. Anti‐GFP
antibody was used in western blot. Protein molecular weight (in kDa) is indicated. Stars indicate the non‐specific bands. Numbers indicate the ratio
of the concentrations of agrobacteria used in co‐infiltration. Empty vector, a binary vector pOCA30 with a 35S promoter; GFP, 35S:GFP in
pOCA30; OsPRI2‐GFP, 35S:OsPRI2‐GFP in pOCA30; OsPRI3‐GFP, 35S:OsPRI3‐GFP in pOCA30; OsHRZ1‐GFP, 35S:OsHRZ1‐GFP in pOCA30.
(e) Cell‐free degradation. Ten‐day‐old roots grown in Fe‐sufficient solution were harvested and used for protein extraction. Incubation with or
without MG132 was performed over the indicated time course
6). SALL4 promotes angiogenesis in gastric cancer by regulating VEGF expression and targeting SALL4/VEGF pathway inhibits cancer progression. Cancer Cell International, 2023
(PubMed: 37525212)
[IF=5.8]
7). Mutated SASH1 promotes Mitf expression in a heterozygous mutated SASH1 knock‑in mouse model. International Journal of Molecular Medicine, 2020
(PubMed: 32582980)
[IF=5.7]
8). Identification and functional analysis of two new de novo KCNMA1 variants associated with Liang–Wang syndrome. Acta Physiologica, 2022
(PubMed: 35156297)
[IF=5.6]
9). Neuropilin-2 Signaling Modulates Mossy Fiber Sprouting by Regulating Axon Collateral Formation Through CRMP2 in a Rat Model of Epilepsy. MOLECULAR NEUROBIOLOGY, 2022
(PubMed: 36044155)
[IF=4.6]
10). ACSS3 regulates the metabolic homeostasis of epithelial cells and alleviates pulmonary fibrosis. Biochimica et biophysica acta. Molecular basis of disease, 2024
(PubMed: 37979225)
[IF=4.2]
Restrictive clause
Affinity Biosciences tests all products strictly. Citations are provided as a resource for additional applications that have not been validated by Affinity Biosciences. Please choose the appropriate format for each application and consult Materials and Methods sections for additional details about the use of any product in these publications.
For Research Use Only.
Not for use in diagnostic or therapeutic procedures. Not for resale. Not for distribution without written consent. Affinity Biosciences will not be held responsible for patent infringement or other violations that may occur with the use of our products. Affinity Biosciences, Affinity Biosciences Logo and all other trademarks are the property of Affinity Biosciences LTD.